
.avif)



Scispot is built to be one of the best LIMS for molecular diagnostics, offering purpose-built workflows for PCR, NGS, and infectious disease testing; integration with leading instruments (e.g. QuantStudio, Roche LightCycler) without custom code; and built-in compliance for CAP, CLIA, ISO 15189, and HIPAA. As Guru Singh, CEO of Scispot, puts it: “We’re building the digital brain for the lab—where every experiment and every sample turns into data that speeds up the next discovery.” Scispot’s molecular diagnostics software centralizes sample tracking, plate design, instrument data, and reporting so labs can accelerate turnaround times and maintain full traceability.
Scispot streamlines laboratory efficiency through comprehensive workflow automation. Our molecular diagnostics software enhances coordination among lab personnel while eliminating manual data entry through instrument integrations. Automated sample tracking reduces search time from nearly an hour to minutes, while built-in quality control checks ensure consistent results. By automating routine tasks, Scispot's molecular diagnostics LIMS allows scientists to focus on analysis rather than paperwork, leading to faster turnaround times and significantly increased sample throughput without additional staffing.
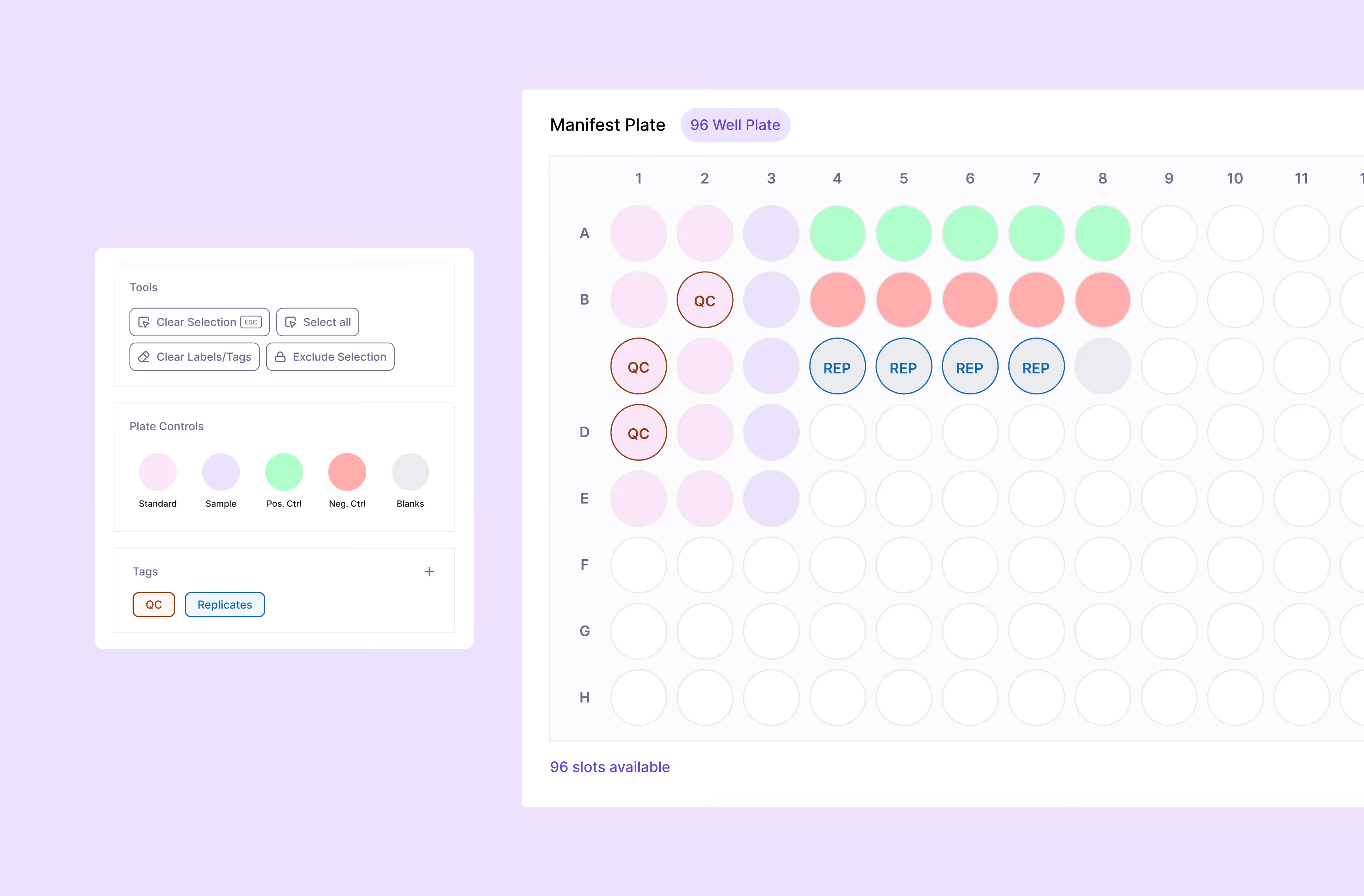
Scispot alt-LIMS supports the full spectrum of molecular diagnostic testing, including PCR/qPCR for infectious disease testing and viral load quantification, next-generation sequencing for oncology gene panels and whole-genome sequencing, and specialized workflows for laboratory-developed tests (LDTs). Our molecular diagnostics software accommodates both high-throughput screening and complex genetic analysis, with customizable workflows for hereditary disease testing, pathogen detection, and pharmacogenomics. The platform's flexibility allows labs to implement both standard assays and novel diagnostic approaches within a single, unified system.
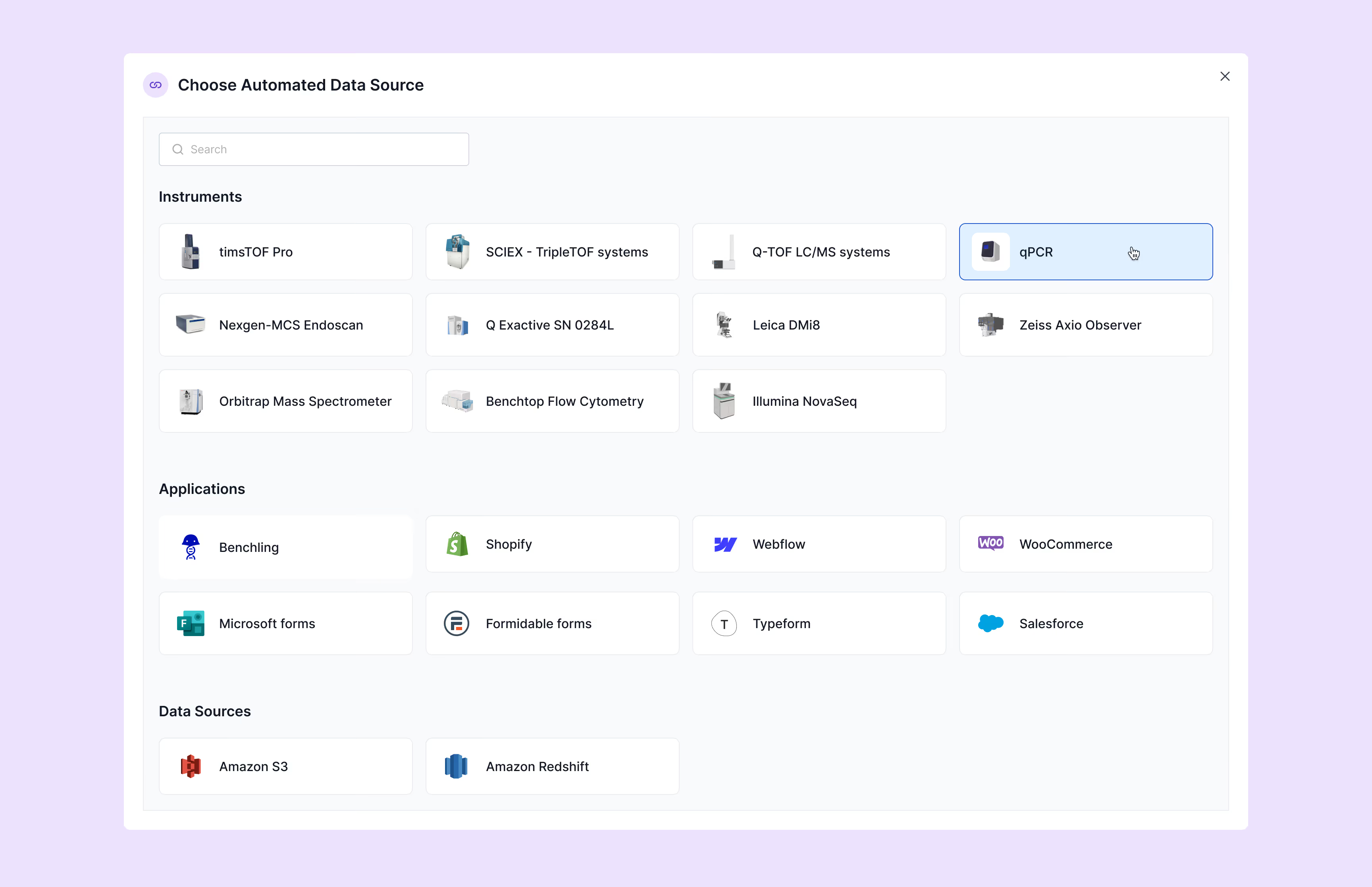
Scispot alt-LIMS integrates seamlessly with your existing ecosystem via HL7 and FHIR standards for LIS/HIS/EHR connectivity. Our molecular diagnostics software features built-in connections with PCR systems, sequencers, and other instruments without requiring custom code. The platform supports both direct instrument interfaces and middleware connections, with flexible API options for specialized equipment. Integration capabilities extend to billing systems and practice management software, creating an end-to-end solution that eliminates information silos and reduces administrative overhead throughout your diagnostic operations.
Scispot alt-LIMS supports LDT workflows with configurable assay designs, validation tracking, and documentation for FDA and CAP requirements. You can define custom test protocols, capture full traceability from sample to result, and maintain audit trails for LDT validation and ongoing QC. The platform’s flexibility allows molecular diagnostics labs to run both FDA-cleared assays and laboratory-developed tests in one system
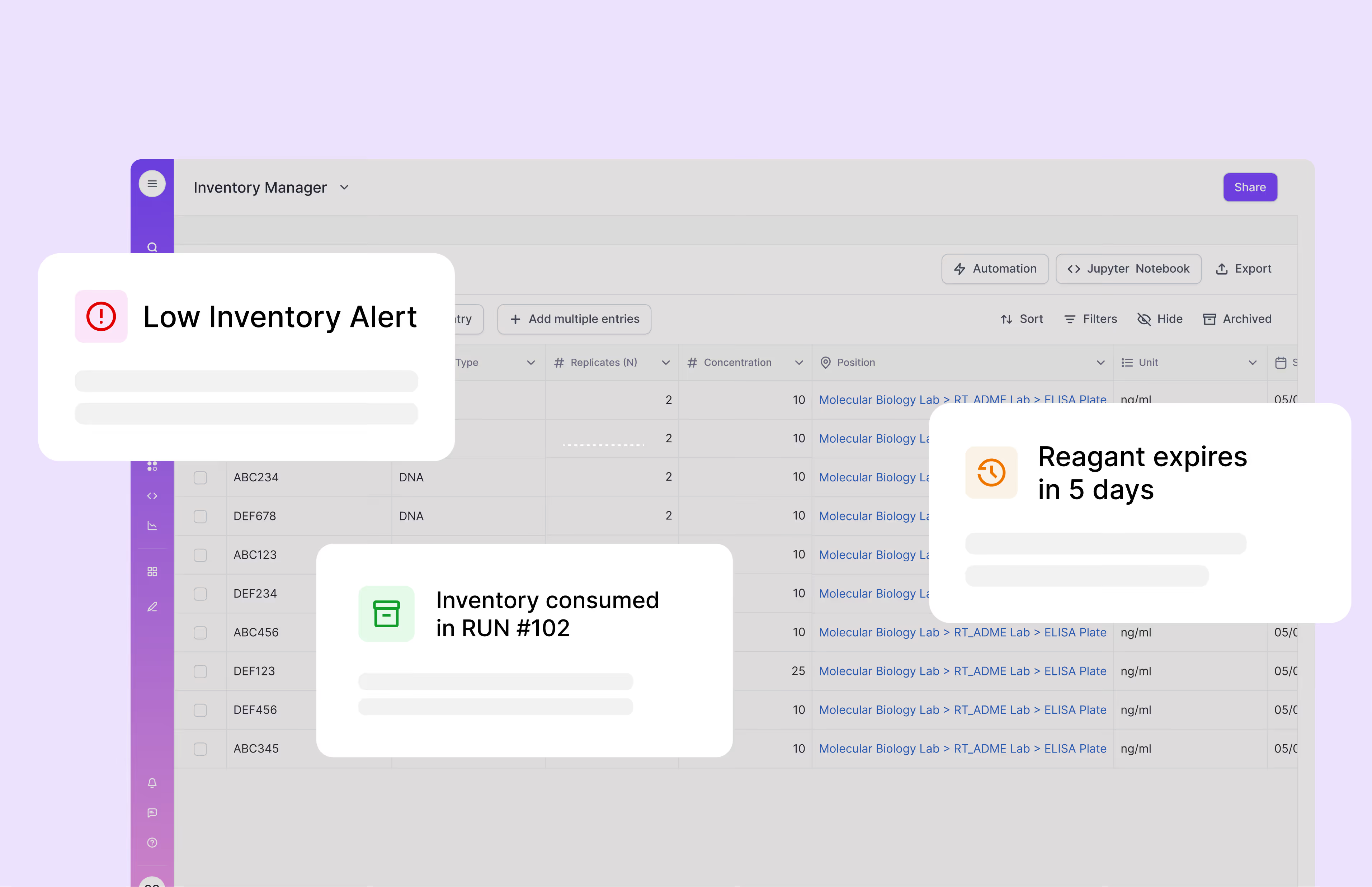
Scispot transforms inventory management with comprehensive tracking for all reagents, kits, and consumables used in molecular testing. Each item is barcoded and monitored in real-time, with automatic documentation of lot numbers and expiration dates to support regulatory compliance. Digital storage mapping shows the exact location of every sample and reagent, dramatically reducing search time. Automated alerts prevent stockouts of critical supplies, while usage analytics help optimize purchasing decisions. This enhanced inventory management has demonstrably reduced waste and prevented testing delays in diagnostic labs of all sizes.